Services
Theragen Bio NGS Service
Theragen Bio,established in 2009 as a research institute within Theragen Etex and has accumulated world-class capabilities over the past 10 years.
We provide genomic analysis services to over 700 medical institutions in Korea and more than 40 research institutions abroad.
In addition, we completed the human genome map In Korea initially and participated in research on the evolution of ancient Ethiopian genomes.Based on our accumulated expertise,Theragen Bio provide Best-in-classNext-Generation Sequencing (NGS) services to our partners.
Click on each button to move to the corresponding page.
- DNA Sequencing
(WGS/Target Seq)
- Transcriptome Sequencing
- Single cell RNA-Sequencing
- Spatial Sequencing
- Epigenome Sequencing
- Metagenome Sequencing
- SNP Array
NGS Sequencing +
Bioinformatics
DNA Sequencing (WGS/Target Seq)
Whole Genome De novo Sequencing
Transcriptome Sequencing
Single-cell RNA Sequencing
GeoMx®
Epigenome Sequencing
Metagenome Sequencing
SNP Array
Global Innovative
Genome Service Provider
Clinical Analysis
Clinical Trial Sample Analysis
FFPE specimen and biomarker discovery
Companion diagnostics development.
1Why Theragen?
Accumulated Technical CapabilitiesSince its establishment as Theragen Bio Research Institute in 2009, we firstly have completed the human genome map as well as
the standard genome maps of the tiger, minke whale, and eagle In 2017, we also received approval as
the first NGS clinical laboratory in Korea from the Ministry of Food and Drug Safety(MFDS).
- No.1
Have No.1 NGS data production and processing system in Korea.(5pb data output in Korea)
- 27
A total of 27 patent registrations, including 10 patent registrations related to large-scale genomic big data.
- 60
Number of service products that have commercialized self-developed products such as customized treatment screening tests, companion diagnostics, liquid biopsy and others.
- 50
Carried out a total of 50 key national tasks, including K-DNA business and healthcare big data constructions
- 150
More than 150 papers published in SCI-class major journals.
- 95
A total of 95 patents
applications, including 3 new
antigen prediction patents
and 5 genomic data patents. - 25%
Secured customers from 700
domestic medical institutions,
occupying a market share of
about 25% in the domesitc
markter, and providing services to customers 40 overseas countries.
-

Good Clinical Laboratory Practice certificate
-

In vitro diagnostic test certificate of clinical laboratory
-

Genetic Testing Accuracy Evaluation Certification
-

ISO9001
-

ISO 27701
-

ISO 27001
2Why Theragen?
Specialized bioinformatics analysisTheragen Bio provides high-quality bioinformatics analysis results, utilizing its expertise in developing the genomic big data service, DEEPOMICS®.
A dedicated manager will be involved throughout the entire process of deriving web-based analysis results.

- DEEPOMICS®, developed by Theragen Bio, is a patient-specific diagnosis and prognosis analysis platform based on AI for genomic big data services. It consists of four platforms: DEEPOMICS® NEO for cancer vaccine research and development, DEEPOMICS® MARKER for analyzing individual patient's concurrent symptoms and prognosis, DEEPOMICS® NETWORK for analyzing patient's drug interactions, and DEEPOMICS® TARGET for selecting effective drug targets for treatment.
-
Theragenbio BI pipeline
-
Analysis Result

3Why Theragen?
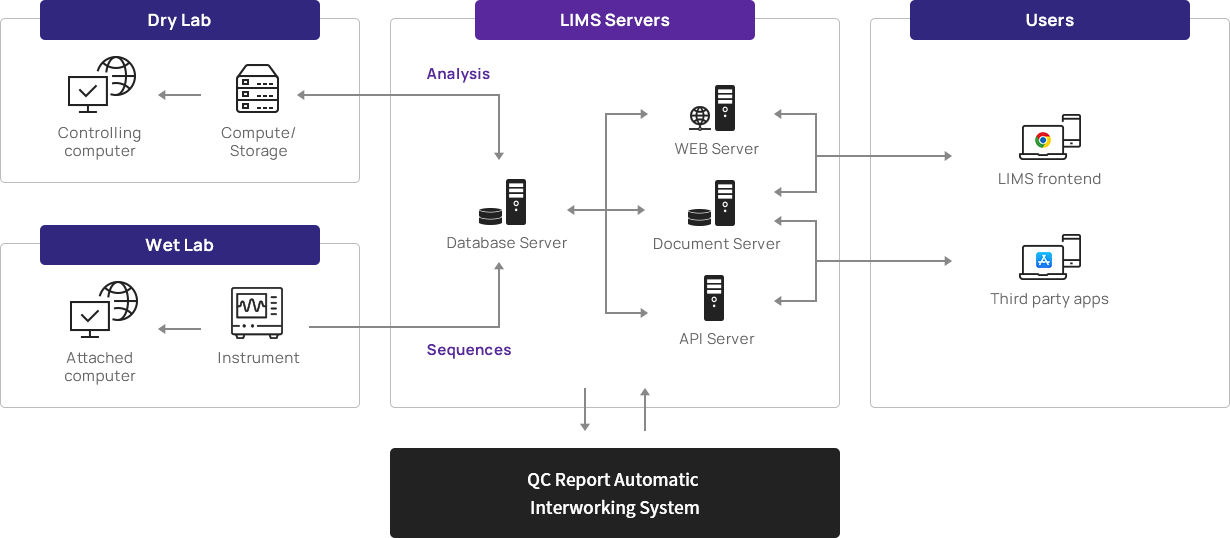
Various and demanding samples Prep/QCWe have our own BIO IT solution (NGS LIMS) and provide high-quality analysis by integrating it in real-time with the entire experimental process.
We have extensive experience in handling various challenging samples, such as specific animal tissues, plants with high levels of polysaccharides, endangered species, and clinical samples with very low amounts of material. Our accumulated experience enables us to provide accurate and reliable results.
Sequencing Platform
| NovaseqX | NovaSeq 6000 | HiSeq 4000 | HiSeq 2500 | |
|---|---|---|---|---|
| Figure |  |  |  |  |
| InstrumentDimension | 34" × 36.7" × 62.5" (86.4cm × 93.3cm × 158.8cm) | 31.5" × 37.2" × 65.2" (80cm × 94.5cm × 165.6cm) | 46.7" × 30" × 37" (118.6cm × 76cm × 94cm) | 46.7" × 30" × 37" (118.6cm × 76cm × 94cm) |
| Workflow | Library Prep > Sequence > Analyze | Library Prep > Sequence > Analyze | Library Prep > Sequence > Analyze | Library Prep> Sequence> Analyze |
| Capable Experiment | Total/Targeted DNA seqWhole Genome Sequencing,Whole Exome seq,Whole-Transcriptome Seq,de novo Seq,Epigenetic regulationRNA-Seq, mRNA-Seq,Amplicon Sequencing,Genotyping by Sequencing,Metagenomics, MeDIP-Seq,ChIP-Seq, Methyl-Seq | WGS,Whole Exome seq,Whole-Transcriptome seq | Next Gen Sequencing,Whole Genome Sequencing,Exome Sequencing,RNA-Seq, mRNA-Seq,Targeted Sequencing,Amplicon Sequencing,Genotyping by Sequencing,Metagenomics, MeDIP-Seq,ChIP-Seq, Methyl-Seq | Exome seq,Whole-Transcriptome Seq,de novo Seq,Epigenetic regulation |
| Explanation | Our new sequencing platform has increased throughput by 2.5 times compared to the previous NovaSeq 6000 while reducing the sequencing time from 48 hours to 24 hours, halving the time required. This increased throughput enables us to generate over 20,000 whole genome sequences per year, surpassing our previous sequencing platform's capacity. | Up to 6Tb and 20 billion cell analyses in 44 hours | 3 billion pairs of Whole genome sequencing in 3.5 days, 180 exosomes in 2 days, or 100 messenger RNA sequencing data can be produced at once. | High-quality data delivery using SBS technology, sequencing equipment that allows flexible use of multiple applications |
| MiSeqDx | Ion S5 XL Sequencer | Ion Proton Sequencer | GeoMx® Digital Spatial Profiler | Sequel Ⅱ | |
|---|---|---|---|---|---|
| Figure |  |  |  |  |  |
| Instrument Dimension | 27" × 22.2" × 20.6" (68.6cm × 56.5cm × 52.3cm) | 8.5" × 22.3" × 22.3" (21.6cm × 56.6cm × 56.6cm) | 21.3" × 30.5" × 18.7" (54.2cm × 77.5cm × 47.4cm) | 30" × 29" × 24" (76.2cm × 73.7 × 61cm) | 72" × 14" × 13" (182.9cm × 35.6cm × 33cm) |
| Workflow | Library Prep > Sequence > Analyze | Library Prep > Sequence > Analyze | Construct library > Prepare templete > Run sequence > Analyze data | Sample Prep > Image and Profile > Count > Analyze | Library Prep > SMRT Sequence > Data Analyze |
| Capable Experiment | Targeted DNA seq | Transcriptome seq,Exome seq,Targeted seq | Agricultural research,Cancer research,Forensic analysis,Human Genome Studies,Stem cell analysis | Whole TranscriptomeSequencing | GS, RNA seq,Targeted seq, complexpopulations,Epigenetics |
| Explanation | A sequencing device designed by combining the SBS technology of Illumina. | Offers a wide range of high-throughput sequencing applications, from microbial genomes, exomes and transcripts | Equipment designed for high sequencing throughput of exosomes, transcriptomes, and genomes | DIGITAL Spatial Profiler equipment capable of analyzing RNA and protein expression and all transcriptome analysis by interlocking with NGS system | Suitable for Whole genome de novo assembly and Full-length 16S rRNA microbiome analysis |
NGS Service Workflow
- 1

-
Consultation and Quotation Issuance
Assignment of dedicated service manager after requesting analysis service
(contact@theragenbio.com)Based on professional service manager consultation
and referral Issuance of an estimate
- 2

-
Sample Submission
Sample submission based on test request form
Packaged sample submission
- 3

-
Sample QC
Providing QC data for the samples
Confirmation of experiment and detailed test request form (TAT confirmation)
Prep QC at the same level as Library QC
Various and rigorous sample Prep QC
- 4

-
Library preparation sequencing
Library preparation tailored to each analysis item
Conducting experiments and
sequencing for optimized results
- 5

-
Bioinformatics analysis
Web-based data analysis through
Theragen Bio's own pipeline
- 6

-
Providing Final Data Analysis Report
Sending analysis result data through
web-based platform
Contact
| Service | Department | Phone number | |
|---|---|---|---|
| NGS | Genome Business | 031-288-1276 | bio@theragenbio.com |
| Bioinformatics | |||
| Healthcare | Healthcare Business |
| bio@theragenbio.com |
| PR/Advertising | PR | 1522-2382 | bio@theragenbio.com |
| Etc. | etc. |




